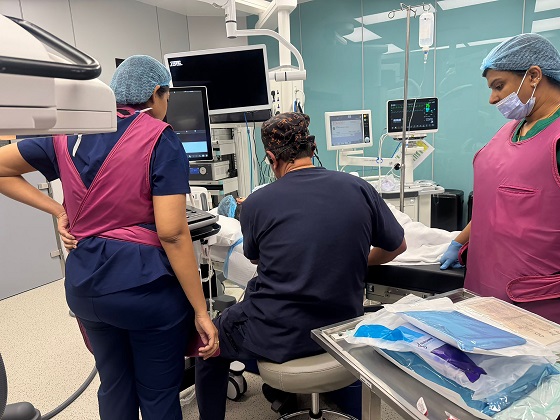
- The 22-year-old Egyptian patient travelled to Dubai to receive the transformative one-time treatment at Medcare Royal Specialty Hospital for his genetic neuromuscular condition. This marks a significant advancement in gene therapy, as this patient is the second adult SMA patient worldwide to be treated with Novartis’ Itvisma as independently verified by the manufacturer Novartis
- Medcare was the first private healthcare provider worldwide to administer the newly licensed Itvisma to a four-year-old international SMA patient last year, also confirmed by the manufacturer Novartis.
- The prevalence of SMA is estimated to be approximately 1 to 2 in 100,000 persons and DMD is estimated to be 1 in 3500 boys worldwide. Since 2020, Medcare has treated over 190 SMA patients and 20 DMD patients with revolutionary gene therapies.

Dubai, UAE – April 28, 2026: Medcare Royal Speciality Hospital (MRSH) in Dubai has become the world’s first hospital outside the USA to offer a newly licensed intrathecal gene therapy to any adult patients with Spinal Muscular Atrophy (SMA). This one-time, life-altering treatment was recently administered to a 22-year-old Egyptian patient who had spent most of his life confined to a wheelchair, with few treatment options available. His case marks a decisive shift in the treatment landscape for SMA, a rare genetic disorder that progressively prevents patients from movement, independence, and, in severe cases, the ability to breathe.